Corresponding author: Tomomi Kotani, itoto@med.nagoya-u.ac.jp
DOI: 10.31662/jmaj.2021-0191
Received: October 18, 2021
Accepted: December 10, 2021
Advance Publication: February 28, 2022
Published: April 15, 2022
Cite this article as:
Kotani T, Imai K, Ushida T, Moriyama Y, Nakano-Kobayashi T, Osuka S, Tsuda H, Sumigama S, Yamamoto E, Kinoshita F, Hirakawa A, Iwase A, Kikkawa F, Kajiyama H. Pregnancy Outcomes in Women with Thyroid Diseases. JMA J. 2022;5(2):216-223.
Introduction: Overt hyperthyroidism and hypothyroidism are associated with pregnancy complications; however, most women with these conditions are diagnosed before conception and are under treatment during pregnancy, especially in high-income countries. The purpose of this study was to investigate pregnancy complications among these women.
Methods: A retrospective cohort study was conducted, and data on pregnant women who gave birth to a singleton at Nagoya University Hospital in Japan in 2005-2014 was collected. The pregnancy outcomes were divided and compared among three groups: the control group (n = 3531), the hyperthyroidism group (n = 48), and the hypothyroidism group (n = 61). Additionally, risk factors for placental abruption were evaluated by multivariable logistic regression analysis. Moreover, in hyperthyroidism, thyroid function at the placentation period was compared between placental-related diseases and nonplacental-related disease groups, and the latter group included placental abruption and preeclampsia.
Results: The incidence of placental abruption was higher in hyperthyroidism than in control and hypothyroidism groups. Hyperthyroidism was independently associated with an increased risk of placental abruption (adjusted odds ratio, aOR = 8.21, 95% confidence interval, CI: 1.76-38.34), as well as preeclampsia (aOR = 4.10, 95% CI: 1.13-14.76) and preterm labor (aOR = 3.38, 95% CI: 1.19-9.64). Additionally, thyroid-stimulating hormone (TSH) at the placentation period was significantly lower in the placental-related disease group than in the nonplacental-related disease group (p < 0.05).
Conclusions: Pregnancy outcomes in women with hyperthyroidism and hypothyroidism would be comparable with those without thyroid disease. Hyperthyroidism was an independent risk factor for placental abruption as well as preterm labor and preeclampsia. However, its frequency was extremely low, and further research is required to validate our findings.
Key words: hyperthyroidism, hypothyroidism, placental abruption, preeclampsia
Thyroid diseases, including hyperthyroidism and hypothyroidism, are common disorders in women of childbearing age. When left untreated, they can have adverse effects on maternal and neonatal outcomes. A large cohort study demonstrated that primary hypothyroidism was associated with increased pregnancy complications, including preeclampsia, gestational diabetes, preterm birth, induction of labor, and cesarean section (1). It also revealed that hyperthyroidism was associated with preeclampsia, preterm birth, and induction of labor. A meta-analysis reported that both overt hypothyroidism and hyperthyroidism were associated with an increase in preterm birth (2). Most women in developed countries with hyperthyroidism or hypothyroidism are diagnosed and treated before conception, and only a few studies have reported the pregnancy outcomes in such cases (3). Thus, before and during pregnancy, the diagnosis and treatment of thyroid disease are recommended to minimize adverse outcomes (4). Previous reports on population-based and case-control studies demonstrated that hyperthyroidism is associated with a high risk of preeclampsia (5), (6), (7), (8) and placental abruption (9). However, the impact of interventions for hyperthyroidism before and during pregnancy on pregnancy outcomes remains uncertain (10), although one study reported a higher occurrence of adverse pregnancy complications in an untreated hyperthyroidism group than a treated group (11). Therefore, even though most women are already on medication, the effect of treated hyperthyroidism or hypothyroidism on pregnancy outcomes remains unknown.
In the present study, we investigated the pregnancy outcomes of women with thyroid disease who had been diagnosed and treated before their pregnancy: birth weight, cesarean section, Apgar scores, and obstetric complications including preterm labor, preeclampsia, light for dates (LFD) infants, premature rupture of membranes (PROM), placenta previa, placenta abruption, and preterm birth. We also focused on placental abruption, the leading cause of maternal and neonatal mortality and morbidity.
The study was approved by the ethics committee of Nagoya University Graduate School of Medicine (2015-0415, 2017-0414). This study was a retrospective study by the ethical guidelines of the Japanese Ministry of Health, Labor, and Welfare. Thus, the requirement for informed consent was waived by the ethics committee of Nagoya University Graduate School of Medicine. However, patients had been informed on the hospital website and provided an option to opt out from their medical records being used in research.
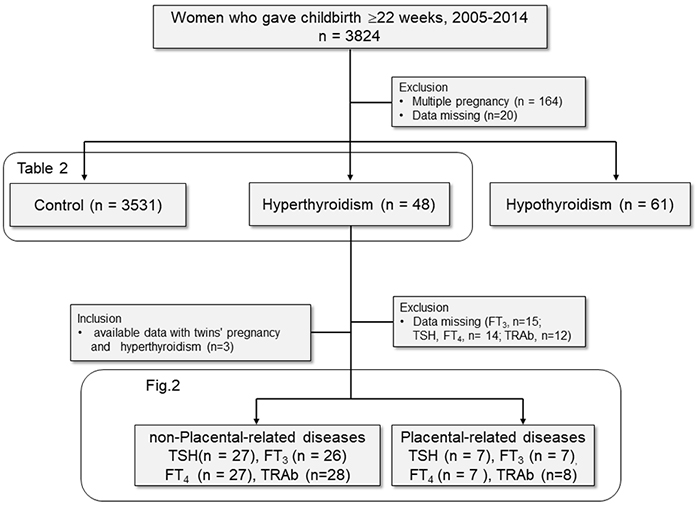
The study samples were retrospectively recruited from medical records at a single tertiary care institute, Nagoya University Hospital in Japan. The inclusion criteria for this study were women who gave birth at Nagoya University Hospital located in Japan from 2005 to 2014 (Figure 1, n = 3824). Additionally, patients who treated thyroid diseases at other hospitals were included when their treatment information from the hospital was provided. The exclusion criteria for this study were as follows: multiple pregnancies (n = 164) and incomplete medical records (n = 20). Subjects were divided into three groups, namely, the control group: those without thyroid disease (n = 3531); the hyperthyroidism group: those who were diagnosed and treated for hyperthyroidism (n = 48); and the hypothyroidism group: those who were diagnosed and treated for hypothyroidism (n = 61). In hyperthyroidism, 21 patients (43.8%) were treated by oral propylthiouracil, and 11 patients (22.9%) were treated by oral methimazole.
Demographic data included maternal age, parity, method of conception, and history of maternal disease, including diabetes mellitus (DM), hypertension, and psychiatric disease. In vitro fertilization and embryo transfer and intracytoplasmic sperm injection were defined as assisted reproductive technology (ART). Additionally, the clinical information on the number of babies, gestational age at delivery, birth weight, fetal position at delivery, mode of delivery, Apgar scores at both 1 and 5 min, and pregnancy complications including preterm labor, preeclampsia, LFD, PROM, placenta abruption, placenta previa, and preterm birth was collected as maternal and neonatal outcomes.
Preeclampsia was defined as persistent hypertension (systolic blood pressure ≥140 mmHg and/or diastolic blood pressure ≥90 mmHg)) and proteinuria (>300 mg/24 h) after 20 weeks of gestation. LFD was defined as birth weight below the 10th percentile, according to a sex-specific Japanese neonatal anthropometric chart (12). Preterm birth was defined as delivery between 22 and 36 weeks of gestation. Gestational ages were calculated on the basis of the last menstrual period and confirmed by crown-rump length when the fertilization date was unknown.
We determined whether hyperthyroidism was an independent risk factor of placental abruption. The crude ORs and 95% CIs of placental abruption were calculated for each of the following factors: maternal age, parity, hyperthyroidism, preeclampsia, preterm labor, placenta previa, and LFD. These are known to be associated with placental abruption (13), (14), (15), (16), (17), (18), (19), (20), (21). Maternal age was analyzed as a continuous variable. The aORs and 95% CIs were calculated using multiple logistic regression analyses, which were performed by adjusting the following variables (p < 0.05): hyperthyroidism, preeclampsia, and preterm labor.
The hyperthyroidism group was further divided into two subgroups (Figure 1); the placental-related disease group: those with preeclampsia or placental abruption and the nonplacental-related disease group: those not complicated with preeclampsia or placental abruption. To use available data as many as possible, we also included twins with hyperthyroidism in the placental-related disease group (n = 2) and the nonplacental-related disease group (n = 1) from the total study population (n = 3824). In the hyperthyroidism group, all patients with preeclampsia developed after 34 weeks of gestation, late-onset preeclampsia. To assess the effect of thyroid function, including stimulating hormone (TSH), free T3 (FT3; triiodothyronine), and free T4 (FT4; thyroxine), and thyroid-stimulating hormone receptor autoantibody (TRAb) positivity on placentation, these were compared in that two subgroups. However, the hyperthyroidism group partially lacked these data because they were treated at other institutes during pregnancy. In the hyperthyroidism group, 34 patients (70.8%) had data of TSH and FT4 at the early gestation, from conception to 15 weeks of gestation, because it is thought to be the period of placental development.
All statistical analyses were performed using the SAS (version 9.4, SAS Institute Inc., Cary, North Carolina, USA) software program and JMP pro 15 (JMP, Tokyo, Japan). Continuous variables are presented as median (range), and p values were calculated using the Mann-Whitney U test and the Kruskal-Wallis test, in comparison with two and three groups, respectively. Categorical variables are presented as numbers (percentage), and p values were calculated by Fisher’s exact test. A p-value of <0.05 was considered to be significant.
Table 1 shows the demographic data of the control (n = 3531), hyperthyroidism (n = 48), and hypothyroidism (n = 61) groups. Regarding conception, a significant difference has been detected among the three groups (p = 0.026), and the percentage of ART in the hypothyroidism group (19.7%) was higher than those of the control (8.9%, p = 0.010). Nevertheless, there were no significant differences in other baseline characteristics like maternal age, nulliparity, and maternal chronic diseases among the control, hyperthyroidism, and hypothyroidism groups.
Table 1. Baseline Characteristics and Pregnancy Outcomes of Pregnancies.
| Control (n = 3531) | Hyperthyroidism (n = 48) | Hypothyroidism (n = 61) | p Value | |
|---|---|---|---|---|
| Baseline characteristics | ||||
| Maternal age, years | 33 (15-52) | 33 (25-42) | 33 (22-43) | 0.102 |
| <20 years | 33 (0.9) | 0 (0.0) | 0 (0.0) | |
| 20-35 years | 2241 (63.5) | 29 (60.4) | 37 (60.7) | 0.907 |
| ≥35 years | 1257 (35.6) | 19 (39.6) | 24 (39.3) | |
| Nulliparity | 1959 (55.5) | 31 (64.5) | 38 (62.3) | 0.262 |
| ART | 314 (8.9) | 4 (8.3) | 12 (19.7) | 0.026* |
| Hypertension | 28 (0.8) | 0 (0.0) | 0 (0.0) | 1.00 |
| DM | 79 (2.2) | 1 (2.1) | 1 (3.3) | 0.661 |
| Psychiatric disease | 190 (5.4) | 1 (2.1) | 4 (6.6) | 0.617 |
| Pregnancy outcomes | ||||
| Gestatinal weeks at birth | 38.7 (22.4-42.6) | 39.4 (33.1-41.3) | 39.3 (32.6-42.0) | 0.015* |
| Birth Weight (g) | 2906 (270-4648) | 2950 (1610-3812) | 3014 (1226-3714) | 0.259 |
| Malposition at delivery | 270 (7.7) | 1 (2.1) | 4 (6.6) | 0.421 |
| Cesarean section | 1461 (41.4) | 17 (35.4) | 25 (41.0) | 0.737 |
| Emergent cesarean section | 590 (16.7) | 10 (20.9) | 13 (21.3) | 0.453 |
| Vacuum or Foreceps | 367 (7.6) | 7 (14.6) | 5 (8.2) | 0.178 |
| Apgar Score ≤6 at 1 min | 368 (10.4) | 2 (4.2) | 1 (1.6) | 0.020* |
| Apgar Score ≤6 at 5 min | 196 (5.6) | 0 (0.0) | 0 (0.0) | 0.031* |
| Preterm labor | 376 (10.7) | 5 (10.4) | 6 (9.8) | 1.000 |
| Preeclampsia | 156 (4.4) | 5 (10.4) | 3 (4.9) | 0.124 |
| LFD infants‡ | 340 (9.7) | 6 (12.5) | 5 (8.3) | 0.708 |
| PROM | 148 (4.2) | 1 (2.1) | 3 (4.9) | 0.796 |
| Placenta previa | 149 (4.2) | 1 (2.1) | 0 (0.0) | 0.264 |
| Placenta abruption | 16 (0.5) | 2 (4.2) | 0 (0.0) | 0.032* |
| Preterm birth | 507 (14.4) | 5 (10.4) | 3 (4.9) | 0.072 |
| Data are presented as number (percentage) and median (range), and p values were calculated by Fisher’s exact and Kruskal-Wallis test, respectively. ‡Nine (Control) cases and one (Hypothyroidism) case were missing data of LFD as over 42 weeks of gestation at delivery.*Statistically significant. ART, assisted reproductive technology; DM, diabetes mellitus; PROM, premature rupture of membranes; LFD, light for dates. | ||||
Pregnancy outcomes were compared among the control, hyperthyroidism, and hypothyroidism groups. No significant differences were observed in the birth weight, fetal malposition at delivery, cesarean section, and vacuum and forceps. However, the incidence of placental abruption was significantly different in the three groups (p = 0.032), and that in the hyperthyroidism group (4.2%) was higher than in the control group (0.5%, p = 0.024). Although the incidence of preeclampsia was not significantly different among the three groups (p = 0.124), the incidence of hyperthyroidism showed a trend to increase when compared with that in the control group (10.4% vs. 4.4%, p = 0.062). The numbers of cases with Apgar scores of ≤6 at 1 and 5 min were significantly different among the control, hyperthyroidism, and hypothyroidism groups (p = 0.020 and p = 0.031, respectively). The percentage of cases with Apgar scores of ≤6 at 1 min in the control group (10.4%) were higher than the hypothyroidism groups (1.6%, p = 0.018). Gestational weeks at birth were earlier in the control group when compared with hyperthyroidism and hypothyroidism groups (p = 0.015). The incidence of preterm birth in the control (14.4%) was also higher than that in the hypothyroidism (4.9%) groups (p = 0.040), but no significant difference was detected among the three groups (p = 0.072). The differences in the other incidences, such as preterm labor, LFD infants, PROM, and placenta previa among the control, hyperthyroidism, and hypothyroidism groups, were insignificant.
Other variables were also examined to determine whether hyperthyroidism was an independent risk factor of placental abruption (Table 2). Factors found to increase placental abruption included hyperthyroidism (crude odds ratio, OR = 9.71, 95% confidential interval, CI: 2.17-43.48), preeclampsia (crude OR = 4.30, 95% CI: 1.23-15.00), and preterm labor (crude OR = 3.26, 95% CI: 1.15-9.20). After multivariate analysis, hyperthyroidism (adjusted OR, aOR = 8.21, 95% CI: 1.76-38.34), preeclampsia (aOR = 4.10, 95% CI: 1.13-14.76), and preterm labor (aOR = 3.38, 95% CI: 1.19-9.64) were found to be independently associated with an increased risk of placental abruption.
Table 2. Univariable and Multivariable Logistic Regression Analysis of Factors Potentially Associated with Placental Abruption.
| Univariable analysis | Multivariable analysis | |||||
|---|---|---|---|---|---|---|
| Crude OR | 95%CI | p value | aOR | 95%CI | p value | |
| Maternal age (years) | 0.957 | 0.869-1.049 | 0.357 | |||
| Nulliparity | 0.793 | 0.314-2.00 | 0.625 | |||
| Hyperthyroidism | 9.71 | 2.17-43.48 | <0.01* | 8.21 | 1.76-38.34 | <0.01* |
| Preeclampsia | 4.30 | 1.23-15.00 | 0.022* | 4.10 | 1.13-14.76 | 0.031* |
| Preterm labor | 3.26 | 1.15-9.20 | 0.025* | 3.38 | 1.19-9.64 | 0.023* |
| Placenta previa | 1.37 | 0.18-10.37 | 0.760 | |||
| LFD infants | 2.68 | 0.88-8.21 | 0.083 | |||
| Maternal age is analyzed as a continuous variable: Its odds ratio is the odds ratio for each additional year of age. OR, odds ratio; CI, confidence interval; aOR, adjusted OR; LFD, light for dates. * Statistically significant. |
||||||
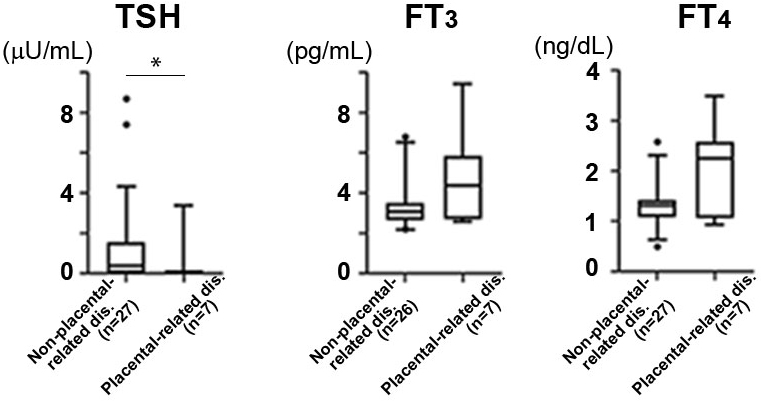
We examined thyroid function at the placentation period, early gestation, to detect specific features in hyperthyroidism that developed placental abruption and preeclampsia. Placental abruption is known to have common pathology as placental-related diseases with preeclampsia (22). In the hyperthyroidism subgroup, serum TSH levels in the placental-related disease group were significantly lower than those in the nonplacental-related disease group (p = 0.043, Figure 2). Although FT3 and FT4 were higher in the placental-related disease group, the difference was insignificant (Figure 2). When comparing TRAb positivity values, no significant difference was detected between the two groups (placental-related diseases―6/8; 75.0%, nonplacental-related diseases―16/28; 57.1%, p = 0.441).
In the present study, pregnancy outcomes in women with hyperthyroidism and hypothyroidism under treatment were comparable with those without thyroid disease. However, the present study revealed that hyperthyroidism had a significantly higher incidence of placental abruption and a trend for increased preeclampsia when compared with the control. Moreover, multivariate analyses showed hyperthyroidism, preeclampsia, and preterm labor as possible risk factors for placental abruption. Additionally, in the hyperthyroidism group, serum TSH levels at the placentation period were lower than in the placental-related disease group, including preeclampsia and placental abruption, and nonplacental-related disease group.
Similar to the present data, hyperthyroidism is associated with a high risk of preeclampsia(5), (6), (7), (8) and placental abruption (9). Additionally, a previous study reported a higher occurrence of hypertensive disorders of pregnancy in the treated group than in control (11), which was consistent with the result in this study because preeclampsia is included in hypertensive disorders of pregnancy.
Additionally, the present study revealed that the maternal and neonatal outcomes in women with treated hypothyroidism were similar to those without thyroid disease. Both overt hyperthyroidism and hypothyroidism are well known for adverse effects on the mother and her child (23). Additionally, ART is known to be associated with increased adverse pregnancy outcomes (24). However, pregnancy outcomes were similar in this study between hypothyroidism and control groups despite a higher prevalence of ART in hypothyroidism. This result is consistent with a previous report (3) and suggests that treatment of hypothyroidism could decrease the risk of adverse pregnancy outcomes seen in overt hypothyroidism.
The recent large cohort reported that the prevalence of placental abruption is as low as 0.5% in Japan (21), which is consistent with the present study. Maternal age, parity, ART, hypertension, DM, psychiatric disease, preeclampsia, preterm labor, LFD, PROM, and placenta previa are well known to be risk factors for placental abruption (13), (14), (15), (16), (17), (18), (19), (20), (21). The present study also showed that preeclampsia and preterm labor were significantly increased in the placental abruption group. However, no significant differences were shown in the other risk factors in the present study. The inconsistency would be related to the small number of the placental abruption group. Multivariate analysis demonstrated that hyperthyroidism, preeclampsia, and preterm labor were independent risk factors for placental abruption in the present study. Several retrospective cohort studies have also reported the association between placental abruption and hyperthyroidism (9), (25). However, whether treatment for hyperthyroidism could influence the risk of placental abruption remains unknown because the present study could not include a population of untreated hyperthyroidism. Furthermore, as randomized controlled trials lacked, the impact of antithyroid interventions for hyperthyroidism prepregnancy or during pregnancy on important pregnancy outcomes remains unknown (9).
Preeclampsia and placental abruption are recently thought to be associated with common pathology as placental-related diseases due to inefficient invasion of trophoblasts after implantation (22). The patients with preeclampsia in the subanalysis were late-onset preeclampsia, and shallow placentation is also detected in late-onset preeclampsia (26). Human chorionic gonadotropin from the trophoblast stimulates thyroid hormones at early gestation. Thyroid hormones play a role in placentation, which is completed at almost 20 weeks of gestation (27), and T3 is known to play a critical role in placentation by increasing trophoblasts invasion (28). These findings led us to hypothesize that thyroid subdysfunction at the placentation period might cause placental-related diseases in hyperthyroidism. To confirm it, we examined TSH levels during the placentation period. They were significantly lower in the placental-related disease group than the nonplacental-related disease group among patients with hyperthyroidism. Serum FT3 and FT4 levels in the placental-related disease group were higher than those in the nonplacental-related disease group, but the difference was insignificant. This thyroid status at the placentation period in the placental-related disease group might be similar to subclinical hyperthyroidism. The previous studies reported the association of placental abruption with subclinical hyperthyroidism at early gestation (25), (29), consistent with the present study. Therefore, these results suggested that treated hyperthyroidism who showed subclinical hyperthyroidism at the placentation period might cause placental-related diseases, including preeclampsia and placental abruption, although further investigation is required. Thus, close check-ups for thyroid function might be concerned at this period in hyperthyroidism. The occurrence of placenta abruption is approximately 0.4% in Japan (30). Therefore, larger cohort is needed to prove it.
The main limitations of this study consisted of the study design being a cohort study and the inclusion of a single tertiary care institution. Prevalence of low Apgar scores and higher preterm births, as well as significantly earlier gestational weeks at birth in the control group when compared with the thyroid disease groups, suggests that the study population belonged to a high-risk category for adverse pregnancy outcomes. Additionally, the general occurrence rate of placental abruption was also low, that is, 0.5%-1.0%. However, the multivariate analysis of the risk of placental abruption might eliminate bias by accounting for confounding factors. Second, all women in the control group were not screened for thyroid dysfunction. Hence, a small but significant proportion of these women could have undiagnosed subclinical thyroid dysfunction, influencing the study results. Subclinical hypothyroidism was associated with preterm birth (7), and these patients may have been included in the control group. Additionally, the information on the time to start treatment, the duration of treatment, and treatment adherence was also lacking, because some of the patients were treated at other hospitals before pregnancy and clinical information before pregnancy was limited. Data on thyroid function were also unavailable for the patients, followed up by other clinics. Most patients treated with methimazole before conception were transitioned into propylthiouracil (n = 7) or reduced (n = 5) during early pregnancy (data not shown). This treatment alternation might cause transient instability of thyroid function but could not be analyzed because of missing data. Further study is needed on the risk of alternation of medication during pregnancy in hyperthyroid patients. Moreover, the sample size for the comparison between the placental-related diseases and nonplacental-related disease groups in hyperthyroidism was small. Further prospective research with a large study population should be conducted on the effect of the intervention before pregnancy on pregnancy outcomes to determine the adequate management of women with hyperthyroidism who wish to be pregnant.
The risks of pregnancy outcomes among women with treated thyroid diseases are comparable with those without thyroid diseases. Although further research is required in a large study population to validate, hyperthyroidism might be an independent risk of placental abruption.
None
This work was supported by JSPS KAKENHI (Grant Number: 15H02660).
We would like to thank Editage (www.editage.jp) for the English language editing.
TK contributed to the conception and design of the study. TK, KI, TU, YM, TNK, HT, and SS performed the acquisition and interpretation of data and revised it critically for important intellectual content. AH and F Kinoshita, as biostatisticians, contributed to the analysis of the data. TK drafted the first version of the manuscript. SO, EY, AI, F Kikkawa and HK contributed to interpreting the data and revising it critically for important intellectual content. All authors gave their approval for the final version of the manuscript.
The study was approved by the ethics committee of Nagoya University Graduate School of Medicine (2015-0415, 2017-0414).
Mannisto T, Mendola P, Grewal J, et al. Thyroid diseases and adverse pregnancy outcomes in a contemporary US cohort. J Clin Endocrinol Metab. 2013;98(7):2725-33.
Sheehan PM, Nankervis A, Araujo Junior E, et al. Maternal thyroid disease and preterm birth: systematic review and meta-analysis. J Clin Endocrinol Metab. 2015;100(11):4325-31.
Matalon S, Sheiner E, Levy A, et al. Relationship of treated maternal hypothyroidism and perinatal outcome. J Reprod Med. 2006;51(1):59-63.
American College of O, Gynecologists. Practice Bulletin No. 148: Thyroid disease in pregnancy. Obstet Gynecol. 2015;125(4):996-1005.
Aggarawal N, Suri V, Singla R, et al. Pregnancy outcome in hyperthyroidism: a case control study. Gynecol Obstet Invest. 2014;77(2):94-9.
Stagnaro-Green A. Overt hyperthyroidism and hypothyroidism during pregnancy. Clin Obstet Gynecol. 2011;54(3):478-87.
Medici M, Korevaar TI, Schalekamp-Timmermans S, et al. Maternal early-pregnancy thyroid function is associated with subsequent hypertensive disorders of pregnancy: the generation R study. J Clin Endocrinol Metab. 2014;99(12):E2591-8.
You SH, Cheng PJ, Chung TT, et al. Population-based trends and risk factors of early- and late-onset preeclampsia in Taiwan 2001-2014. BMC Pregnancy Childbirth. 2018;18(1):199.
Pillar N, Levy A, Holcberg G, et al. Pregnancy and perinatal outcome in women with hyperthyroidism. Int J Gynaecol Obstet. 2010;108(1):61-4.
Earl R, Crowther CA, Middleton P. Interventions for hyperthyroidism pre-pregnancy and during pregnancy. Cochrane Database Syst Rev. 2013;(11):Cd008633.
Wang Y, Sun XL, Wang CL, et al. Influence of screening and intervention of hyperthyroidism on pregnancy outcome. Eur Rev Med Pharmacol Sci. 2017;21(8):1932-7.
Itabashi K, Miura F, Uehara R, et al. New Japanese neonatal anthropometric charts for gestational age at birth. Pediatr Int. 2014;56(5):702-8.
Tikkanen M. Placental abruption: epidemiology, risk factors and consequences. Acta Obstet Gynecol Scand. 2011;90(2):140-9.
Matsuda Y, Hayashi K, Shiozaki A, et al. Comparison of risk factors for placental abruption and placenta previa: case-cohort study. J Obstet Gynaecol Res. 2011;37(6):538-46.
Hasegawa J, Nakamura M, Hamada S, et al. Capable of identifying risk factors for placental abruption. J Matern Fetal Neonatal Med. 2014;27(1):52-6.
Nagata C, Yang L, Yamamoto-Hanada K, et al. Complications and adverse outcomes in pregnancy and childbirth among women who conceived by assisted reproductive technologies: a nationwide birth cohort study of Japan environment and children’s study. BMC Pregnancy Childbirth. 2019;19(1):77.
Ananth CV, Oyelese Y, Srinivas N, et al. Preterm premature rupture of membranes, intrauterine infection, and oligohydramnios: risk factors for placental abruption. Obstet Gynecol. 2004;104(1):71-7.
de Paz NC, Sanchez SE, Huaman LE, et al. Risk of placental abruption in relation to maternal depressive, anxiety and stress symptoms. J Affect Disord. 2011;130(1-2):280-4.
Zhong QY, Gelaye B, Fricchione GL, et al. Adverse obstetric and neonatal outcomes complicated by psychosis among pregnant women in the United States. BMC Pregnancy Childbirth. 2018;18(1):120.
Anderson E, Raja EA, Shetty A, et al. Changing risk factors for placental abruption: a case crossover study using routinely collected data from Finland, Malta and Aberdeen. PLoS One. 2020;15(6):e0233641.
Kyozuka H, Murata T, Fukusda T, et al. Teenage pregnancy as a risk factor for placental abruption: findings from the prospective Japan environment and children’s study. PLoS One. 2021;16(5):e0251428.
Younis JS, Samueloff A. Gestational vascular complications. Best Pract Res Clin Haematol. 2003;16(2):135-51.
Laurberg P, Andersen SL. Endocrinology in pregnancy: pregnancy and the incidence, diagnosing and therapy of Graves’ disease. Eur J Endocrinol. 2016;175(5):R219-30.
Luke B. Pregnancy and birth outcomes in couples with infertility with and without assisted reproductive technology: with an emphasis on US population-based studies. Am J Obstet Gynecol. 2017;217(3):270-81.
Zhang Y, Li Y, Shan Z, et al. Association of overt and subclinical hyperthyroidism during weeks 4-8 with adverse pregnancy outcomes. J Womens Health (Larchmt). 2019;28(6):842-8.
Stanek J. Histological features of shallow placental implantation unify early-onset and late-onset preeclampsia. Pediatr Dev Pathol. 2019;22(2):112-22.
Silva JF, Ocarino NM, Serakides R. Thyroid hormones and female reproduction. Biol Reprod. 2018;99(5):907-21.
Vissenberg R, Manders VD, Mastenbroek S, et al. Pathophysiological aspects of thyroid hormone disorders/thyroid peroxidase autoantibodies and reproduction. Hum Reprod Update. 2015;21(3):378-87.
Arbib N, Hadar E, Sneh-Arbib O, et al. First trimester thyroid stimulating hormone as an independent risk factor for adverse pregnancy outcome. J Matern Fetal Neonatal Med. 2017;30(18):2174-8.
Ohira S, Motoki N, Shibazaki T, et al. Alcohol consumption during pregnancy and risk of placental abnormality: the Japan environment and children’s study. Sci Rep. 2019;9(1):10259.